Inside access: Arman Sarshoghi
Cryoablation protocols for primary and metastatic lung tumors
Cryoablation Protocols for Primary and Metastatic Lung Tumors: A Systematic Review and Meta-Analysis Evaluating Effectiveness and Safety of Percutaneous Cryoablation of Pulmonary Tumors. Sarshoghi, Arman et al. Journal of Vascular and Interventional Radiology, Volume 37, Issue 1, 107871
Tell us about you, your team, and your institution.
Arman Sarshoghi: Our team is based at the Centre Hospitalier de l'Université de Montréal (CHUM), affiliated with Université de Montréal, though this work truly represents an international collaboration with co-authors from Johns Hopkins, Memorial Sloan Kettering, the Chinese University of Hong Kong and Thoraxklinik Heidelberg.
Our lab, the Technology, Innovation and Development Lab (TID Lab), led by Moishe Liberman, MD, PhD, takes a translational approach to medical device development. We identify physician-led gaps in available technology and move innovations from prototyping through ex-vivo human organ testing, live animal studies and ultimately first-in-human clinical trials. We also collaborate with established medical device companies and start-ups to help develop, validate and test their devices. This project exemplifies that philosophy: identifying a clinical gap (the lack of standardized cryoablation protocols) and systematically working to address it through rigorous evidence synthesis.

Why did you pursue this topic?
Sarshoghi: This topic emerged directly from our clinical experience. Dr. Liberman is a thoracic surgeon with deep expertise in interventional pulmonology, and we see tremendous potential for cryoablation to evolve beyond its current percutaneous applications; particularly toward transbronchial ablation, which could enable same-day biopsy and treatment for select patients.
However, as we reviewed the literature to inform our own practice and future device development, we were struck by how little evidence underpins current protocols. The prescribed freeze-thaw cycles, durations and target temperatures vary dramatically from center to center, often based on tradition or manufacturer recommendations rather than rigorous data. We felt that before we could advance the modality, we needed to understand what the existing evidence tells us and where the critical gaps lie.
What are the key takeaways from your research?
Sarshoghi: Three main findings stand out. First, the extreme heterogeneity in clinical practice. Cryoablation protocols vary enormously across centers: different numbers of freeze-thaw cycles, different durations, different endpoints. This inconsistency limits our ability to compare outcomes and improve the technique systematically.
Second, while the pooled 1-year local tumor control rate of 90.5% is encouraging, cryoablation is often reserved as a last-line treatment for patients who cannot undergo surgery or who have lungs too fibrotic for stereotactic body radiation therapy (SBRT). This positioning may undersell its potential.
Third, and perhaps most importantly, unlike SBRT or microwave ablation, lung cryoablation completely lacks the fundamental biology and physics framework that would allow evidence-based dosimetry. We have well-established dose-response relationships for radiation (DNA damage and Gray) and heat ablation (protein denaturation and thermal dose), but nothing comparable for cryotherapy (see Figure 1). This is a critical gap that must be addressed if we want to optimize outcomes and expand indications.
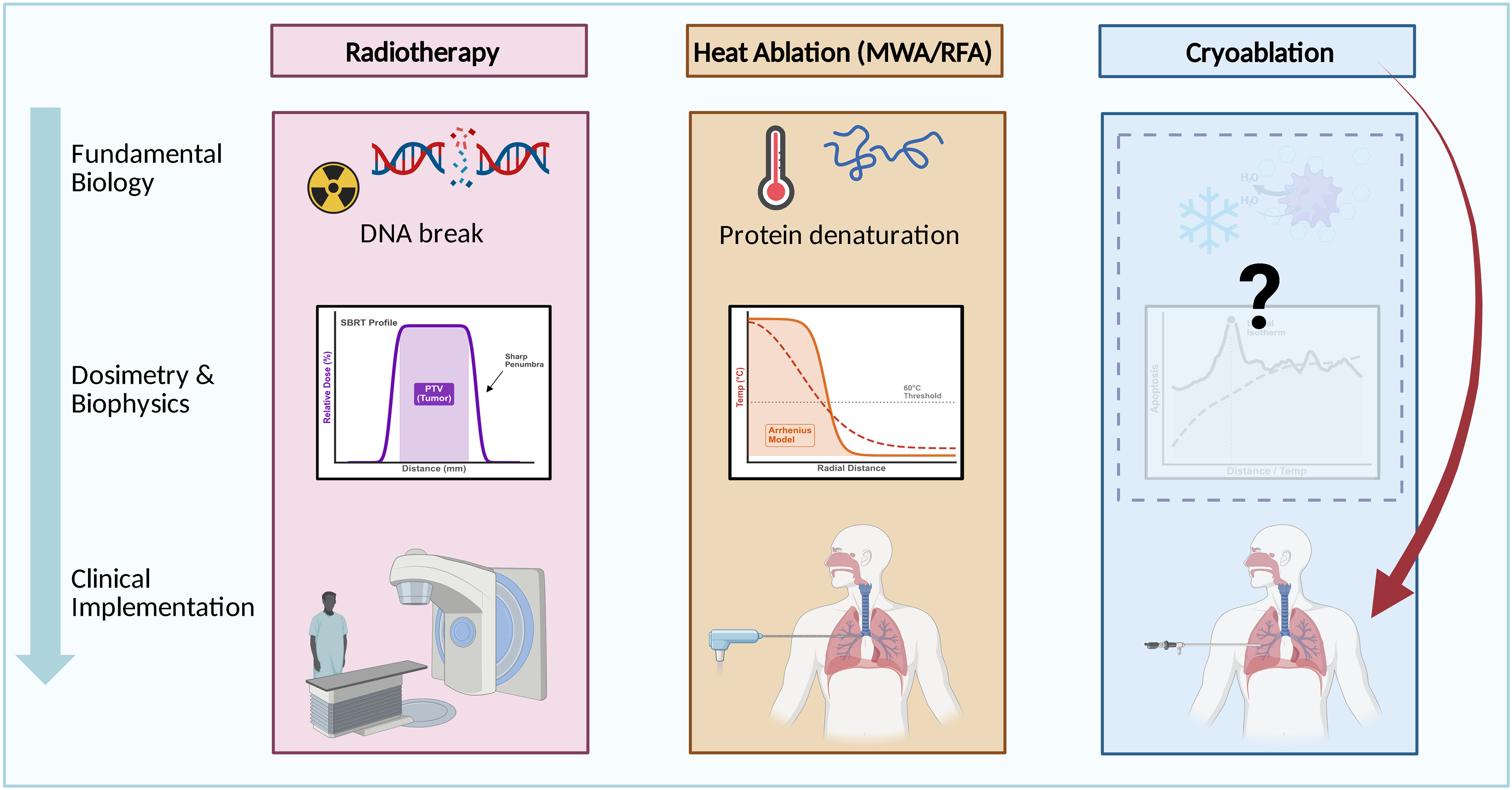
How might this research influence treatment, practice or clinical processes in interventional radiology?
Sarshoghi: In the near term, this meta-analysis provides practitioners with data-driven guidance on protocol optimization; specifically, the value of triple freeze-thaw cycles with a short initial freeze followed by longer subsequent freezes. For clinicians currently using double-cycle protocols, this offers a concrete, evidence-based reason to reconsider their approach.
More broadly, we hope this work catalyzes a shift toward standardized reporting and eventually standardized dosimetry for cryoablation. If we can establish reliable dose-response relationships, we open the door to patient-specific treatment planning, similar to what radiation oncology achieved decades ago.
Looking further ahead, our ultimate vision is to enable same-day diagnosis and treatment via transbronchial approaches: navigate to a lesion bronchoscopically, obtain a biopsy, confirm malignancy and ablate. Achieving this safely and effectively requires the kind of evidence-based dosimetry framework this research begins to establish.
What do you see as the most important clinical takeaway from this meta-analysis on lung cryoablation, and how might it influence everyday practice for interventional radiologists?
Sarshoghi: The most important takeaway is recognizing both the heterogeneity and the opportunity it represents. Current practice varies so widely that there is clearly room for optimization.
For everyday practice, interventional radiologists should consider adopting triple freeze-thaw protocols if they haven't already, with attention to the sequencing we identified: a shorter initial "priming" freeze (~3–5 minutes) followed by longer subsequent freezes (~7–10 minutes), ending with a thaw cycle. This isn't just adding a cycle, it's a different procedural philosophy with mechanistic rationale.
But beyond specific protocol changes, we hope this work encourages the IR community to push for better dosimetry. Cryoablation is an incredible modality, it offers real-time visualization, preserves collagenous architecture and has a favorable safety profile, yet it's being left behind compared to other ablative technologies because we lack the fundamental framework to optimize and standardize it. That needs to change.
The study suggests improved outcomes with a triple freeze–thaw protocol and specific freeze durations. How do these findings align with your current approach, and do they prompt any changes in how you perform lung cryoablation?
Sarshoghi: These findings largely validate and refine what many experienced practitioners have been gravitating toward empirically. The triple freeze-thaw protocol has been gaining traction based on earlier work by Hinshaw and others showing enhanced cytotoxicity and our meta-analysis provides population-level evidence supporting this approach.
What's particularly interesting is the specific timing pattern that emerged: a short initial freeze followed by progressively longer freezes. The mechanistic explanation makes sense, the brief first freeze induces localized hemorrhage and alters the lung's thermal properties, essentially "priming" the tissue for more effective subsequent freezes with larger ablation zones and more circular isotherms.
In our own work, we've moved toward this approach, and we're now conducting laboratory studies to develop true dose-response frameworks that link thermal distribution profiles to tumor kill. This is where the field needs to go from empirical protocol optimization to physics-based treatment planning.
Any next steps or plans for follow up research?
Sarshoghi: Absolutely. This meta-analysis was really the first step in a larger research program aimed at establishing evidence-based dosimetry for cryoablation.
We're currently conducting laboratory studies using clinical-grade cryoprobes to develop true dose-response curves. We've built custom experimental platforms that allow us to precisely map, model and simulate temperature fields during freeze-thaw cycles while simultaneously tracking tumor viability. Our goal is to quantify the relationship between thermal exposure and tumor kill and essentially create the equivalent of radiation dose-response curves, but for cryotherapy.
We are also developing computational tools that could enable patient-specific treatment planning based on tumor geometry and location. The aim is to provide the scientific foundation for transbronchial cryoablation, where precise dosimetry becomes even more critical given the constraints of working through the airways with smaller probes. We believe this approach could eventually enable same-day biopsy and ablation—a true paradigm shift in how we manage early-stage lung malignancies.
