SIR Today
First-in-human results on RF denervation for pancreatic cancer pain
Presentation: Sunday, April 12 at 3 p.m. during the Pain Management/MSK 1 session.
Schwartz R, Sabirov A, Bedoya M, Machan L
A new first-in-human proof-of-concept study has shown that RF denervation of the somatosensory nerve can improve pain and quality of life while reducing opioid use.
“Our abstract reports findings from a first-in-human proof-of-concept study evaluating transvascular radiofrequency (RF) denervation of the celiac plexus in patients with refractory pancreatic cancer–related abdominal pain,” said researchers from AKFA Medline Hospital in Uzbekistan, the University of British Columbia, and Autonomix Medical, Inc. “The goal was to assess procedural feasibility, safety and early clinical outcomes.”
Autonomix is developing a catheter-based sensing and RF ablation platform designed to improve how physicians identify and treat peripheral nerves.
“Today, many neuromodulation procedures are performed without direct confirmation that the intended neural targets have been effectively treated, which can lead to variability in outcomes,” the team said.
Pancreatic cancer pain was selected as their initial indication due to the high prevalence of severe, neuropathic pain and the significant unmet need for improved treatment options. According to researchers, approximately 70% of pancreatic tumors involve nerve structures, and up to 75% of patients experience moderate to severe pain. In addition, this indication allows for smaller, focused early-stage clinical studies, which is appropriate for first-in-human evaluation of a novel approach.
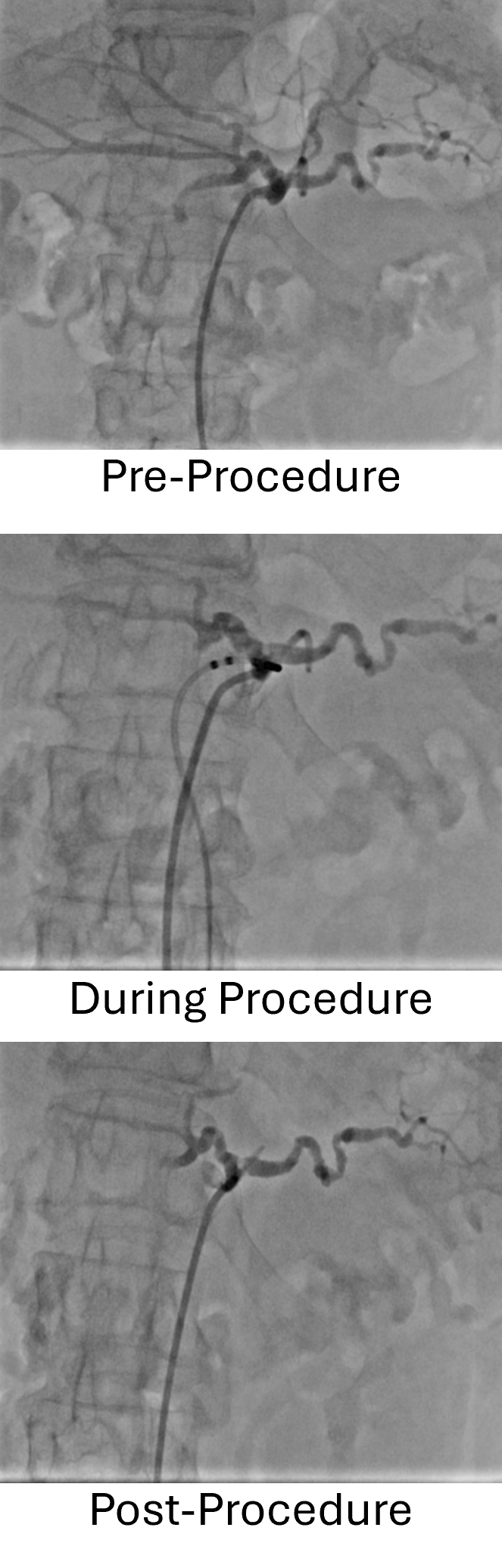
In this prospective study, 19 of 20 enrolled patients underwent successful treatment, corresponding to a 95% procedural feasibility rate, researchers said. This total population group observed significant reductions in pain as early as 24 hours following the procedure, with mean pain reductions exceeding 40% through three months of follow-up. Importantly, there were no device- or procedure-related serious adverse events.
“100% of subjects who underwent a femoral access procedure responded to treatment,” researchers said. That group had a 59% reduction in pain, and 100% of responding patients were opioid-free within 7 days. At 4-6 weeks, 73% remained opioid free.
“We also learned that patients with brachial access did not respond, marking an important procedural finding,” they said. “These results provide early clinical evidence that transvascular neuromodulation can be performed safely and may meaningfully reduce severe pancreatic cancer–related pain.”
While the proof-of-concept study was small and exploratory, the consistency of pain relief and favorable safety profile support further investigation, researchers said. These findings also provide proof-of-concept for transvascular neuromodulation more broadly and establish a foundation for integrating electrophysiological sensing into future procedures to improve precision and procedural confidence.
“This study represents an early but important step in validating a new transvascular approach to neuromodulation,” the team said. “The goal at Autonomix is to enable minimally invasive, data-informed nerve-targeted therapies that provide greater procedural control and have the potential to improve patient outcomes.”
As the field of catheter-based neuromodulation evolves, technologies that combine treatment with functional confirmation may help advance precision in nerve-targeted interventions, they said, and these early results are a key indicator of value.
“These results provide early clinical evidence that transvascular neuromodulation can be performed safely and may meaningfully reduce severe pancreatic cancer–related pain,” they said.

